Formation of Ionic bonds and Properties
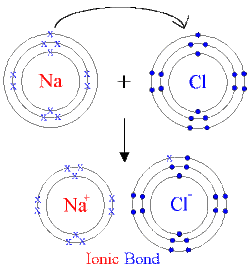
Ionic bond is formed with a metal and nonmetal.
For example:Sodium and fluorine bonding ionically to form sodium fluoride. Sodium loses its outer electron to give it a stable electron configuration, and this electron enters the fluorine atom exothermically. The oppositely charged ions are then attracted to each other, and their bonding releases energy. The net result is that energy leaves the atoms, so the reaction is able to take place.
Source: wikipedia.org
For example:Sodium and fluorine bonding ionically to form sodium fluoride. Sodium loses its outer electron to give it a stable electron configuration, and this electron enters the fluorine atom exothermically. The oppositely charged ions are then attracted to each other, and their bonding releases energy. The net result is that energy leaves the atoms, so the reaction is able to take place.
Source: wikipedia.org
